New Electronic Transplantation RecordFeedback Form
To our valued customers,
You may be aware that the FDA and the American Association of Tissue Banks (AATB) both have requirements regarding the traceability of allograft tissue, which are tissue products produced from deceased human donors. Under 21 Code of Federal Regulations Chapter I, Subchapter L, Part 1271.290(f)1, a federal regulation, tissue establishments (tissue banks), such as AlloSource, are required to maintain systems that ensure the ability of the tissue bank to trace a piece of allograft tissue to the patient who receives the transplanted tissue, or to discard. Similarly, facilities (hospitals, surgery centers, etc.) which use allografts must also establish their own systems to ensure traceability of the transplanted or discarded tissue, per The Joint Commission’s regulations.
As a customer of AlloSource, your facility purchases allografts which are used in various medical procedures performed at your facility.
Currently AlloSource includes a paper-based Transplant Record & Feedback Form (TRFF), the document that allows us to maintain traceability, in each allograft package to assist facilities in complying with the requirements. The form requires the allograft’s lot number, information as to whether the tissue was implanted or discarded and on what date, and the facility information.
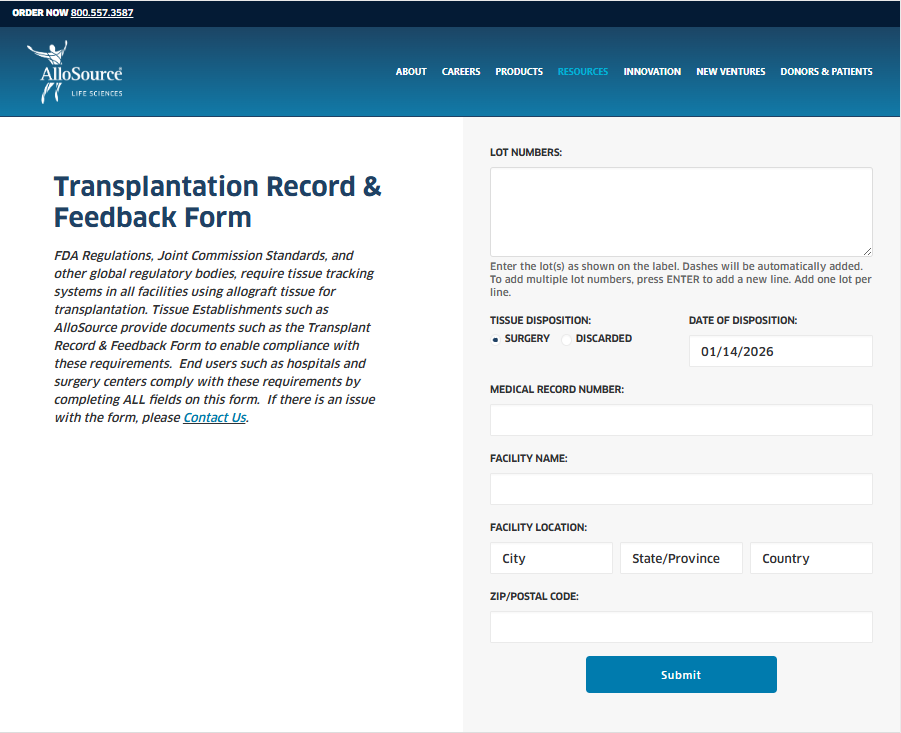
However, paper forms are cumbersome for both the facility staff (time to fill out and return) and AlloSource (issues with legibility). To help improve compliance, AlloSource is transitioning to an insert which states the regulatory requirements and provides a QR code, which directs to an online TRFF form. The same form can also be accessed directly at www.allosource.org/TRFF. This web address will also be printed on the insert.
You can view a screenshot of the online form in Attachment A, and review the insert with the QR code in Attachment B.
For a period of time, product packages may contain the insert with no QR code, as we transition out of products which have the current form, and into those which have the QR code. However, you may use the link at www.allosource.org/TRFF beginning on January 9, 2026.
At the same time, we will discontinue including our Terms and Conditions in each shipment. The web address to a link on our website where the Terms and Conditions can be viewed will be printed on every pack slip. It can be viewed at any time by visiting https://allosource.org/resources/terms-and-conditions/.
Please pass this information along to any others who will find it useful. If you have questions, please reach out to your AlloSource representative.
Thank you in advance for continuing to help us fulfill our obligations under the federal regulations, upholding your own responsibilities, and assisting us in honoring the gift of all donors.
Sincerely,
Your partners at AlloSource
1 Title 21 of the Code of Federal Regulations 1271.290(f) states that tissue establishments must establish and maintain a method for recording the distinct identification code and type of each HCT/P distributed to a consignee to enable tracking from the consignee to the donor; establish and maintain a method for documenting the disposition of each of your HCT/Ps,to enable tracking from the donor to the consignee or final disposition; and inform consignees, in writing, of the traceability requirements and of their (tissue establishment) tracking systems employed to ensure compliance.
Online Form
Transplantation Record Feedback Form Insert




